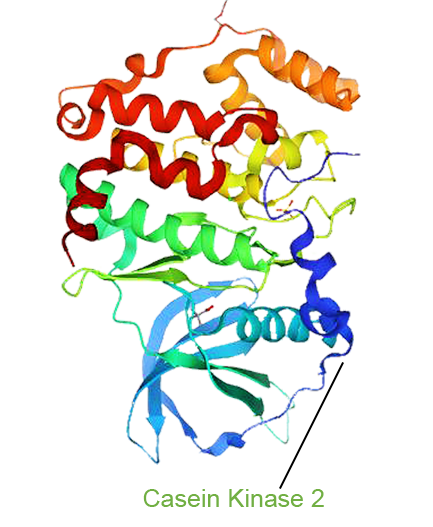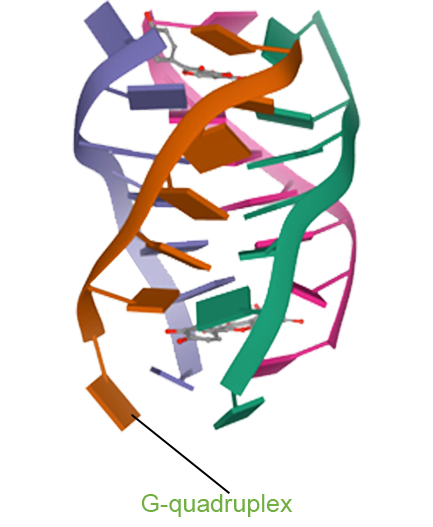- Bringing Hope to Life
- Senhwa is dedicated to developing first-in-class drugs to address unmet medical needs, bringing new hope to patients. Currently, the company is advancing multiple innovative programs, including a five-year cancer research project sponsored by the U.S. National Cancer Institute (NCI), focusing on monotherapy, combination cancer immunotherapy (IO), and antibody-drug conjugates (ADC). Additionally, Senhwa focuses on cancers caused by specific gene deficiencies, rare diseases, and abnormal inflammatory responses linked to viral infections.
- OUR PIPELINE

- Google AI–Validated Immuno-Modulator
- Silmitasertib (CX-4945) has been validated by Google DeepMind’s latest biological AI model, emerging from a pool of 4,000 candidate drugs. It demonstrates immune-reprogramming capability by enhancing antigen presentation, converting immunologically “cold” tumors into “hot” tumors. This breakthrough reveals a novel therapeutic pathway for cancer and positions Silmitasertib as a highly promising next-generation backbone drug for combination cancer therapies.
- Platform
- Turn Cold Tumors into Hot Ones
- Pidnarulex (CX-5461) has demonstrated the potential to convert “cold tumors” into “hot tumors” by altering the tumor microenvironment, thereby enhancing the therapeutic response to immune checkpoint inhibitors (ICIs). Preclinical studies confirm that Pidnarulex induces immunogenic cell death, activating immune cells and improving the tumor microenvironment. When combined with immunotherapy, Pidnarulex holds promise for treating a variety of cancers resistant to ICI monotherapy.
- PUBLICATIONS

- Development of Innovative Orphan Drugs
- Silmitasertib (CX-4945) has obtained four orphan drug designations, including for cholangiocarcinoma, biliary tract cancer, medulloblastoma, and neuroblastoma. It has also received rare pediatric disease designation for the treatment of medulloblastoma and neuroblastoma. The future market launch will benefit from advantages such as tax credits and a seven-year exclusive market period in the United States.
- OUR CLINICAL TRIALS